✔ 100% Authentic Product
👁️ Currently Viewing 1993
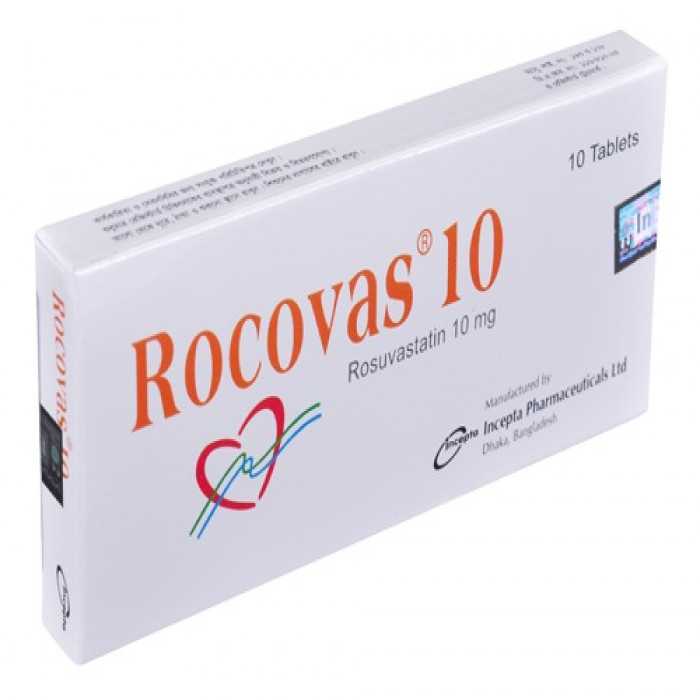
Tablet, Generic Name: Rosuvastatin, Manufacturer/Distributor: Drug International Ltd.
Discount
Price: ৳ 106
MRP:
৳
112
5%
Off

100% Genuine Products, Guaranteed

Safe & Secure Payments, Always

Fast, Secure & Efficient Delivery
Proper Packaging
 Cash on Delivery - All over Bangladesh
Cash on Delivery - All over Bangladesh Regular Delivery - 12-24 Hours, Dhaka City* Charge Tk.39-59
Regular Delivery - 12-24 Hours, Dhaka City* Charge Tk.39-59 Regular Delivery - 24-48 Hours, Other Cities* Charge Tk.99-110
Regular Delivery - 24-48 Hours, Other Cities* Charge Tk.99-110
 ফ্রি ডেলিভারিঃ - ৯৯৯ টাকা+ অর্ডারে, ঢাকা
শহরে
ফ্রি ডেলিভারিঃ - ৯৯৯ টাকা+ অর্ডারে, ঢাকা
শহরে ফ্রি ডেলিভারিঃ - ২৯৯৯ টাকা+ অর্ডারে, ঢাকার
বাহিরে
ফ্রি ডেলিভারিঃ - ২৯৯৯ টাকা+ অর্ডারে, ঢাকার
বাহিরে
100% Genuine Products, Guaranteed
Safe & Secure Payments, Always
Fast, Secure & Efficient Delivery
Proper Packaging
 Cash on Delivery - All over Bangladesh
Cash on Delivery - All over Bangladesh Regular Delivery - 12-24 Hours, Dhaka City* Charge Tk.39-59
Regular Delivery - 12-24 Hours, Dhaka City* Charge Tk.39-59 Regular Delivery - 24-48 Hours, Other Cities* Charge Tk.99-110
Regular Delivery - 24-48 Hours, Other Cities* Charge Tk.99-110 ফ্রি ডেলিভারিঃ - ৯৯৯ টাকা+ অর্ডারে, ঢাকা
শহরে
ফ্রি ডেলিভারিঃ - ৯৯৯ টাকা+ অর্ডারে, ঢাকা
শহরে ফ্রি ডেলিভারিঃ - ২৯৯৯ টাকা+ অর্ডারে, ঢাকার
বাহিরে
ফ্রি ডেলিভারিঃ - ২৯৯৯ টাকা+ অর্ডারে, ঢাকার
বাহিরে
✅ Description:
Primary hypercholesterolemia (type IIa including heterozygous familial hypercholesterolemia), mixed dyslipidemia (type IIb), or homozygous familial hypercholesterolemia in patients who have not responded adequately to diet and other appropriate measures; prevention of cardiovascular events in patients at high risk of a first cardiovascular event.
Dosage: Before treatment initiation the patient should be placed on a standard cholesterol-lowering diet that should continue during treatment. The dose should be individualized according to the goal of therapy and patient response, using current consensus guidelines.
Treatment of hypercholesterolemia: Patient of Asian origin or with risk factors for myopathy or rhabdomyolysis: initially 5 mg once daily increased if necessary to max. 20 mg daily.
Prevention of cardiovascular events: Patient of Asian origin or with risk factors for myopathy or rhabdomyolysis: initially 5 mg once daily increased if necessary to max. 20 mg daily.
Pediatric Use (Hyperlipidemia including familial hypercholesterolemia):
- Child younger than 6 years: not recommended.
- Child 6–9 years: initially 5 mg daily, increased if necessary at intervals of at least 4 weeks to usual max. 10 mg once daily.
- Child 10–18 years: initially 5 mg daily, increased if necessary at intervals of at least 4 weeks to usual max. 20 mg once daily.
[Reduced dose required with concomitant atazanavir, darunavir, ezetimibe, fibrate, itraconazole, lopinavir, or tipranavir]
Use in the elderly (>70 years): A start dose of 5 mg is recommended. No dose adjustment necessary.
Renal insufficiency: Initially 5mg once daily (do not exceed 20 mg daily) if eGFR is 30-60 mL/minute/1.73 m2. Avoid if eGFR is less than 30 mL/minute/1.73 m2
Hepatic impairment:
- Child-Pugh scores of <7: no increase in systemic exposure to rosuvastatin.
- Child-Pugh scores of 8 and 9: increased systemic exposure has been observed. In these patients an assessment of renal function should be considered.
- Child-Pugh scores >9: no study.
Rosuvastatin is contraindicated in patients with active liver disease.
Race: Increased systemic exposure has been seen in Asian subjects. The recommended starting dose is 5 mg for patients of Asian ancestry. The 40 mg dose is contraindicated in these patients.
Genetic polymorphisms: Specific types of genetic polymorphisms are known that can lead to increased rosuvastatin exposure. For patients who are known to have such specific types of polymorphisms, a lower daily dose of Rosuvastatin is recommended.
Dosage in patients with pre-disposing factors to myopathy: The recommended starting dose is 5 mg in patients with predisposing factors to myopathy. The 40 mg dose is contraindicated in some of these patients.
Side effects: Common or very common: Proteinuria.
Rare: Hepatitis, jaundice.
Very rare: Gynecomastia, hematuria, hepati failure, interstitial lung disease, lupus erythematosus-like reactions, pancreatitis.
Frequency not known: Alopecia, altered liver function tests, amnesia, arthralgia, asthenia, depression, dizziness, edema, fatigue, gastrointestinal disturbances, headache, hypersensitivity reactions, hyperglycemia -may be associated with the development of diabetes mellitus (particularly in those already at risk of the condition), myalgia, myopathy, myositis, paresthesia, peripheral neuropathy, pruritus, rash, rhabdomyolysis, sexual dysfunction, sleep disturbance, Stevens-Johnson syndrome, thrombocytopenia, urticaria, visual disturbance.
Muscle effects: The risk of myopathy, myositis, and rhabdomyolysis associated with statin use is rare. Although myalgia has been reported commonly in patients receiving statins, muscle toxicity truly attributable to statin use is rare. Muscle toxicity can occur with all statins, however the likelihood increases with higher doses If muscular symptoms or raised creatine kinase occur during treatment, other possible causes (e.g. rigorous physical activity, hypothyroidism, infection, recent trauma, and drug or alcohol addiction) should be excluded before statin therapy is implicated, particularly if statin treatment has previously been tolerated for more than 3 months. When a statin is suspected to be the cause of myopathy, and creatine kinase concentration is markedly elevated (more than 5 times upper limit of normal), or if muscular symptoms are severe, treatment should be discontinued. If symptoms resolve and creatine kinase concentrations return to normal, the statin should be reintroduced at a lower dose and the patient monitored closely; an alternative statin should be prescribed if unacceptable side-effects are experienced with a particular statin. Statins should not be discontinued in the event of small, asymptomatic elevations of creatine kinase. Routine monitoring of creatine kinase is unnecessary in asymptomatic patients.
Statins should not be discontinued if there is an increase in the blood-glucose concentration or HbA1C as the benefits continue to outweigh the risks.
Interstitial lung disease: If patients develop symptoms such as dyspnoea, cough, and weight loss, they should seek medical attention.
⚠️Disclaimer:
At ePharma, we’re committed to providing accurate and accessible health information. However, all content is intended for informational purposes only and should not replace medical advice from a qualified physician. Please consult your healthcare provider for personalized guidance. We aim to support, not substitute, the doctor-patient relationship.